Nucleic Acid Aptamer Selection Using Beta-glucuronidase for Monitoring Gene Expression
Sarah
Kaddour-Djebbar, Fall 2013
Abstract
Aptamers are oligonucleic acids
or peptide molecules that bind to a specific target molecule and are utilized
as biomolecular recognition units. Aptamers are usually created by selecting
them from a large random sequence pool. Nucleic acid aptamers are single
stranded ssDNA or RNA. Selection is carried out through many oligonucleotides
that have a central region of variable size and random sequence and two
flanking regions of known sequence to allow for PCR amplification. Aptamers
have been engineered through repeated rounds of in vitro selection to specifically
bind to various targets, such as proteins, small molecules, nucleic acids, and
even cells and tissues. They are capable of identifying a target molecule with
high affinity and specificity, which allows for potential as diagnostic tools. Application
of aptamers is also used for both basic research and clinical purposes as
macromolecular drugs. Beta-glucuronidases, shown in Figure 1, are members of
the glycosidase family of enzymes that catalyze the breakdown of complex
carbohydrates. Beta-glucuronidase has a molecular weight of 57,000 g/mol. It functions as a negatively charged tetrameric protein
with an isoelectric charge of 4.8. Human beta-glucuronidase is located in the
lysosome. In the gut, beta-glucuronidase converts conjugated bilirubin to the
unconjugated for reabsorption. Human beta-glucuronidase is similar to the E.
coli enzyme beta-galactosidase.
Beta-glucuronidase can have
effects on a patient’s health as well as other medically related factors. Deficiencies
in beta-glucuronidase result in the non-recessive inherited metabolic disease
known as Sly syndrome. This results in the build-up of non-hydrolyzed mucopolysaccharides
in a patient. This disease can be extremely debilitating for the patient or can result in hydrops fetalis, which is a condition in the fetus characterized by an accumulation of fluid prior to birth. In addition, mental retardation, short stature, coarse facial features, spinal abnormalities, and enlargement of liver and spleen are observed in surviving patients. A certain amount of beta-glucuronidase
activity appears to be important for normal enterohepatic recirculation of
endogenous compounds such as vitamin D, thyroid hormone, and estrogen. Elevated
beta-glucuronidase can deconjugate potential toxins, increasing the formation
of carcinogens in the bowel and promoting the enterohepatic recirculation of
toxins, hormones, and various drugs in the body. Research correlates elevated
levels of beta-glucuronidase with increased colon cancer risk. In fact,
excessive beta-glucuronidase activity may be a primary factor in the etiology
of colon cancer. Beta-glucuronidase can break down epithelium cells and can
cause the spread of cancer, specifically colon cancer. There is an aptamer that
can bind and inhibit beta-glucuronidase which can detect cancer in the
epithelium cells.
Successfully
isolating an aptamer that can locate beta-glucuronidase could help doctors
detect colon cancer by observing levels of beta-glucuronidase in the body.
Specific Aim 1
The primary specific aim of this
project focuses on generating a specific aptamer the can bind to GUS-His.
Specific Aim 2
The next specific aim is using beta-glucuronidase
to monitor gene expression for colon cancer.
Figure 1: Beta-glucuronidase binding with the target proteinBeta-glucuronidases are members of the glycosidase family of enzymes that catalyze breakdown of complex carbohydrates. Human β-glucuronidase is synthesized as an 80 kDa monomer (653 amino acids) before proteolysis removes 18 amino acids from the C-terminal end to form a 78 kDa monomer. Beta-glucuronidase exists as a 332 kDa homotetramer. Beta-glucuronidase contains several notable structural formations, including a type of beta barrel known as a jelly roll barrel and a TIM barrel.
View the full proposal here.
View the first progress report here.
View the second progress report here.
View the final report here.
View the full proposal here.
View the first progress report here.
View the second progress report here.
View the final report here.
Target Order Information
Vendor: Ellington Lab
Vendor Website: https://www.ellingtonlab.org
Vendor Telephone Number:
Central Lab Telephone 512-471-6445
Office Manager 512-232-3426
Cost per Unit: $0
Cost per Round: $0
References
Sinnott, Michael, ed. (1998). Comprehensive Biological Catalysis. Manchester, UK: Academic Press. pp. 119-138.
Nyhan, William L.; Bruce Barshop, Pinar Ozand (2005). Atlas of Metabolic Diseases (2 ed). London, UK: Hodder Arnold. pp. 501-503, 546-550.
Henrissat B, Bairoch A (1993). "New families in the classification of glycosylhydrolases". Biochem J 293: 781-788.
Henrissat B (1991). "A classification of glycosyl hydrolases based on amino-acid sequence similarities". Biochem J 280: 309-316.
Sinnott, Michael, ed. (1998). Comprehensive Biological Catalysis. Manchester, UK: Academic Press. pp. 119-138.
Nyhan, William L.; Bruce Barshop, Pinar Ozand (2005). Atlas of Metabolic Diseases (2 ed). London, UK: Hodder Arnold. pp. 501-503, 546-550.
Henrissat B, Bairoch A (1993). "New families in the classification of glycosylhydrolases". Biochem J 293: 781-788.
Henrissat B (1991). "A classification of glycosyl hydrolases based on amino-acid sequence similarities". Biochem J 280: 309-316.
Adlercreutz H, Martin F, Pulkkinen
M, et al. Intestinal metabolism of estrogens. J Clin Endocrin Metab
1976;43(3):497-505.
Kuhn JG. Pharmacology of
irinotecan. Oncology 1998;12(8 Suppl 6):39-42.
Reddy BS, Wynder EL. Large-bowel
carcinogenesis: Fecal constituents of populations with diverse incidence rates
of colon cancer. J Natl Cancer Insti 1973;50(6):1437-42.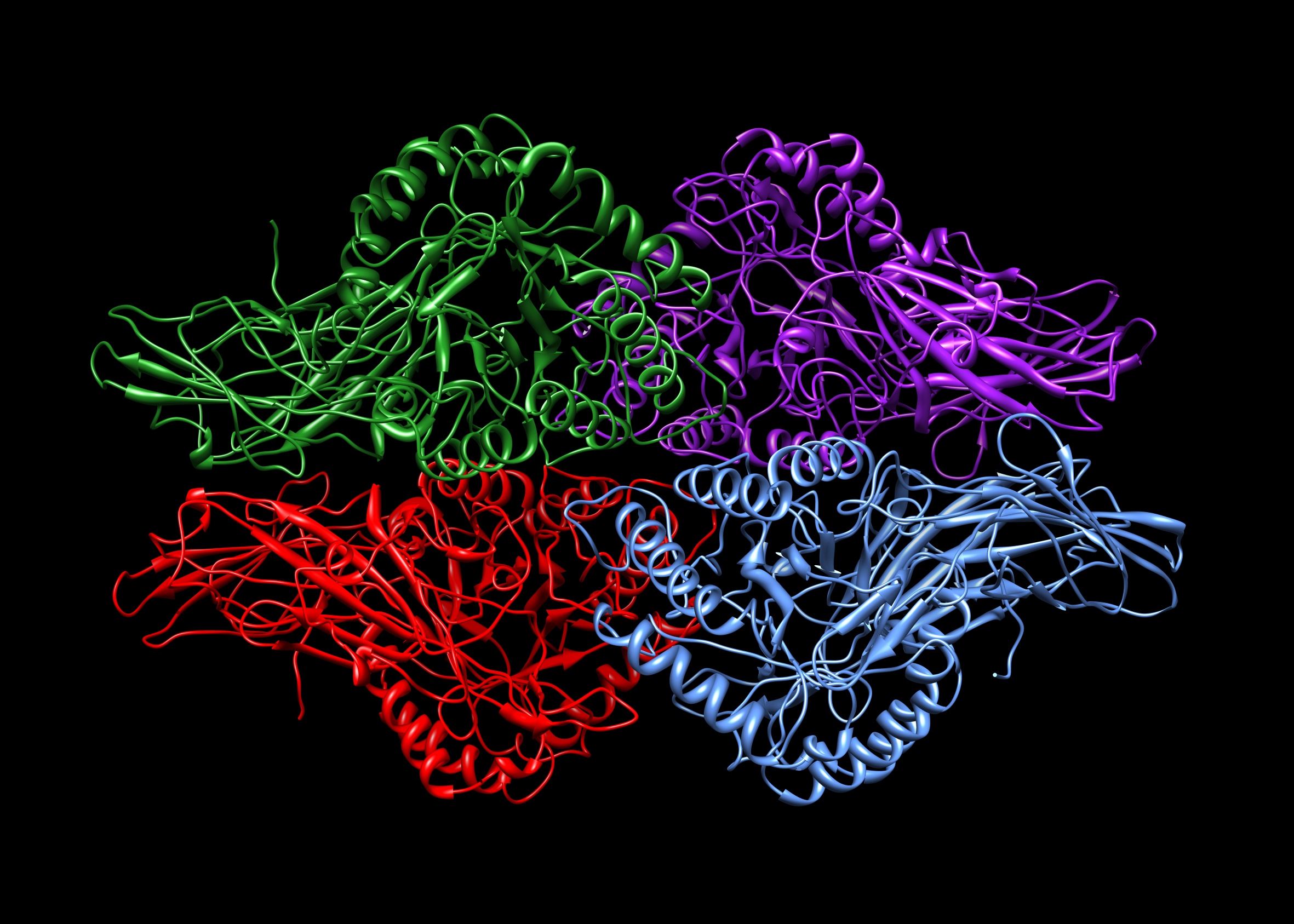
No comments:
Post a Comment